Available online at: https://doi.org/10.18778/1898-6773.87.3.07
 https://orcid.org/0009-0007-1799-4784
https://orcid.org/0009-0007-1799-4784
Department of Anthropology, Manipur University
 https://orcid.org/0000-0002-2914-0264
https://orcid.org/0000-0002-2914-0264
Department of Anthropology, Manipur University
ABSTRACT: Urolithiasis is one of the most prevalent urinary diseases worldwide. Several studies have reported VDR gene polymorphisms to have a contributing genetic factor in susceptibility to urolithiasis and suggested its possibility of being a good candidate marker for urolithiasis. However, results across numerous studies centred on the relationship between the VDR gene polymorphism and urolithiasis have been inconclusive. Therefore, we performed a meta-analysis concerning the association between the risk of urolithiasis and VDR gene polymorphisms viz., ApaI, BsmI, FokI, and TaqI among the Asian population. A comprehensive electronic search was conducted to identify published studies that investigates the relationship between four polymorphisms (ApaI, BsmI, FokI and TaqI) in the VDR gene and the risk of urinary stone disease using electronic databases. VDR ApaI and FokI polymorphisms were found to be associated with urolithiasis risk. Results from pooled analysis indicated ApaI aa genotype to be associated with urolithiasis compared to AA or Aa genotypes. In addition, the minor f allele of FokI variant was identified to be the risk allele in susceptibility to urolithiasis while F allele to be protective. Moreover, from the subgroup analysis, the ff genotype of FokI and aa genotype of ApaI were associated with higher risk of urolithiasis among the East Asian but not among the Southwest Asians.
KEY WORDS: Vitamin D receptor, genetic polymorphism, SNPs, urinary stones disease, allele, genotype.
Urolithiasis is one of the most prevalent urinary diseases worldwide, causing a huge burden on healthcare systems. It is a complex disease that may result from various factors, such as environment, diet, age, genetic factors, metabolic disorders (Fallahzadeh et al. 2012). Urolithiasis is characterized by a high recurrence rate and its incidence continues to increase (Lee et al. 2002). Gene polymorphism has been reported to be an important factor associated with the disorder. A growing amount of epidemiological studies have highlighted the allelic variation in the Vitamin D Receptor (VDR) gene to be involved in the urinary stone disease.
The human VDR, which is a member of the steroid receptor family, is a product of a single gene and is located on chromosome 12 at 12q13-14, and it comprises 11 exons that, together with intervening 8 introns, span approximately 75 kb (Arababadi et al. 2011; Labuda et al. 1992; Miyamoto et al. 1997). Single nucleotide polymorphisms (SNPs) of the VDR gene, commonly ApaI (rs7975232), BsmI (rs1544410), FokI (rs2228570) and TaqI (rs731236) have been intensively studied to investigate associations between these polymorphisms and the risk of urolithiasis as they have been hypothesized to influence the expression and/or function of the VDR protein (Subaşı et al. 2017). These are the restriction enzymes widely used to study the VDR gene polymorphism. Among the four VDR variants, three of them (ApaI, BsmI and TaqI) occur in the intron sections, while only the FokI variant changes the codon (González-Castro et al. 2019; Ou et al. 2014), located at the translational start site of exon 2 of the VDR gene, has been reported to alter the VDR protein sequence and associated with a reduced response to vitamin D in target cells (Arai et al. 1997). Whereas, the SNPs ApaI and BsmI located in exon 8 and TaqI in exon 9 have also been proven to enhance messenger RNA (mRNA) stability or transcriptional activity, and therefore they may lead to an increase in vitamin D activity (Morrison et al. 1994). Several investigations have observed VDR gene polymorphisms to have a contributing genetic factor in susceptibility to urolithiasis and suggested its possibility of being a good candidate marker for urolithiasis (Bid et al. 2005; Chen et al. 2001a; Jawad and Awad 2020). In this light, a well-established genetic marker would have a profound influence in screening and preventing urolithiasis. However, results across numerous studies centred on the relationship between the VDR gene polymorphism and urolithiasis have been inconclusive.
Previous meta-analyses (Imani et al. 2020; Lin et al. 2011; Yang et al. 2019; Zhou et al. 2013) studied the Asian population as a subgroup and their conflicting results could have been due to the ethnic diversity across the region. Hence, our study aims to perform a meta-analysis looking at the the association between the risk of urolithiasis and VDR gene polymorphisms, viz., ApaI, BsmI, FokI, and TaqI among the Asian population specifically, and to further analyze by stratifying them based on regional variation into East Asians and the Southwest Asians. The present meta-analysis is expected to provide a better understanding and may clarify the association between the VDR gene polymorphism and urolithiasis among the Asian populations.
Search strategy: A comprehensive electronic search was conducted (from December 2023 upto March 2023) to identify all published studies that have investigated the relationship between four polymorphisms (ApaI, BsmI, FokI and TaqI) in the VDR gene and the risk of urinary stone disease. Three electronic databases, Science Direct, PubMed and Google Scholar, were searched. The databases were searched using the combination of the following keywords: ‘Urolithiasis’, ‘Nephrolithiasis’, ‘Kidney stones’, ‘VDR gene polymorphism’, ‘SNP’.
Inclusion criteria: (1) The data supplied the frequency of each genotype or the raw data for computing the frequency of each genotype, which could calculate odd ratios and 95% confidence intervals (CIs). (2) Studies documenting the link between VDR polymorphisms and urolithiasis susceptibility. (3) Case-control studies
Exclusion criteria: (1) Studies with preliminary results not on VDR BsmI, Fok1, TaqI and ApaI gene polymorphism or outcome. (2) Duplicate data, case reports, book chapters, reviews, letters, and articles with no full text or detailed data were excluded. There was a language limit, and studies not available in English were excluded.
Two investigators independently screened the literature search and retrieved the titles, abstracts, and full texts of articles that matched the search terms. Then, the same authors cross checked the selections made and any disagreement between the authors were resolved by consensus. The first author’s name, journal and year of publication, country of origin, ethnicity, mean or range of age, and total sample size of cases and controls and for each gender separately, the VDR variant studied and genotype frequencies were extracted. The Newcastle-Ottawa Scale (NOS) was applied to assess the quality of relevant articles. The NOS evaluation criteria included three aspects: selection of research subjects (4 points), comparability of research subjects (2 points) and risk factor exposure (3 points) consisting of of nine points in total. The total score of each article could be rated from 0 (lowest) to 9 (highest). The quality of the article was considered to be low if the score was less than 5 points and high if the score was more than or equal to 5 points (Luchini et al. 2017). Quality assesments were performed by the two authors independently and any discrepancies in the evaluations were resolved by discussion or by the assistance of a third researcher if needed.
The association between the risk of urolithiasis and the gene polymorphisms of VDR ApaI, BsmI, FokI, and TaqI was determined by the calculation of pooled odds ratios (OR) and 95% confidence intervals (CI). For pooled OR to be statistically significant, p<0.05 was required. Between studies heterogeneity was estimated using Cochrane’s Q test, significant at p<0.05. Besides, I2 was used to quantitatively report the heterogeneity among the included studies. In absence of heterogeneity, the pooled statistic was performed using the fixed effects model, but a random effects model was conducted when the p-value of the heterogeneity test was <0.10. All data were analyzed using Metagenyo (Martorell-Marugan et al. 2017) to calculate the available data from each study. Seven statistical models were applied in this meta-analysis, viz. Allelic model which compares the minor allele to major allele (ApaI a vs. A; BsmI b vs. B; FokI f vs. F and Taq t vs. T); Recessive model (ApaI aa vs. AA+Aa; BsmI bb vs. BB+Bb; FokI ff vs. FF+Ff; and TaqI tt vs. TT+Tt), Dominant model (ApaI Aa+aa vs. AA; BsmI Bb+bb vs. BB; FokI Ff+ff vs. FF; and TaqI Tt+tt vs. TT), Over-dominant model (ApaI Aa vs. aa+AA; BsmI Bb vs. bb+BB; FokI Ff vs. ff+FF and TaqI Tt vs. tt+TT), and three pair-wise comparison models viz. Homozygous model (ApaI aa vs. AA, BsmI bb vs. BB, FokI ff vs. FF, and TaqI tt vs. TT), heterozygous model (ApaI Aa vs. AA, BsmI Bb vs. BB, FokI Ff vs, FF and TaqI Tt vs.TT) and minor allele homozygote versus heterozygote model (Apa aa vs. Aa, BsmI bb vs. Bb, FokI ff vs. Ff and TaqI tt vs. Tt). In addition, subgroups based on the region in Asia, viz., East Asia and Southwest Asia were stratified.
We used Egger’s test for an objective evaluation of the publication bias. If the P-value of Egger’s test is greater than 0.05 and the funnel plot is symmetrical, it can be considered that there is no significant evidence of publication bias. We also performed a sensitivity analysis to assess whether individual studies affected the overall results by leave one out method. The analysis is repeated after removing one study each time, and it was found that the combined odds ratio (OR) of the remaining studies was within the 95% CI in the meta-analysis. The results showed that the combined OR of this meta-analysis had good stability.
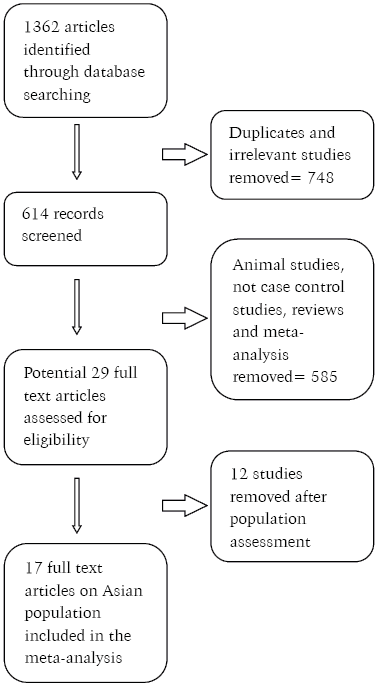
Study characteristics: Figure 1 outlines the study selection process. A total of 1362 articles were identified after the initial search from the electronic databases. After an initial screening of titles and abstracts using inclusion and exclusion criteria, 29 studies were identified for full-text review, and 17 articles were included in quantitative analysis.

Fig. 1. Flowchart illustrating the study selection process
A total of 17 aticles reporting the relationship between VDR ApaI, BsmI, FokI, and TaqI gene variants and urolithiasis susceptibility were recruited through the search strategy. The required data from these studies were extracted (Tab. 1). Of these 17 articles, 6 articles studied the ApaI (rs7975232) variant, 4 articles for BsmI (rs1544410) variant, 11 articles for FokI (rs2228570) variant, and 10 articles for TaqI (rs731236) variant.
| Reference | Age group | Stone composition | VDR polymorphic site | Cases | Controls | NOS |
| Amar et al., (2020) | NA | NA | BsmI,FokI, TaqI, | 235 | 243 | 7 |
| Bid et al., (2005a) | Adult | Calcium oxalate | FokI | 138 | 166 | 6 |
| Bid et al., (2005b) | Children | NA | FokI | 50 | 60 | 8 |
| Chen et al., (2001a) | Adult | Calcium oxalate | FokI | 146 | 90 | 5 |
| Chen et al., (2001b) | Adult | Calcium oxalate | BsmI | 124 | 90 | 4 |
| Guha et al., (2015) | Adult | Calcium Stones | FokI, TaqI | 200 | 200 | 8 |
| Huang et al., (2019) | Children | NA | FokI | 142 | 238 | 7 |
| Jawad & Awad, (2020) | All | Calcium Stones | TaqI | 204 | 127 | 5 |
| Liu et al., (2007) | Adult | Calcium Stones | FokI | 235 | 231 | 8 |
| Mittal et al., (2010) | Adult | NA | ApaI,FokI, TaqI | 125 | 150 | 7 |
| Nishijima et al., (2002) | Adult | Calcium Stones | ApaI, TaqI | 83 | 86 | 6 |
| Parvaresh et al., (2022) | Children | Calcium Stones | TaqI | 90 | 90 | 7 |
| Seo et al., (2010) | Adult | Calcium Stones (major) | ApaI,FokI, TaqI | 273 | 525 | 7 |
| Relan et al., (2004) | Adult | Calcium oxalate (major) | BsmI, FokI | 150 | 100 | 7 |
| Shaogang et al., (2003) | Adult | Calcium oxalate | ApaI,FokI, TaqI | 150 | 80 | 6 |
| Wang et al., (2012) | Adult | Calcium Stones | ApaI,BsmI, FokI, TaqI | 464 | 450 | 8 |
| Yang et al., (2019) | Adult | Calcium oxalate | ApaI, TaqI | 943 | 975 | 8 |
NOS: New Castle Ottawa Scale
NA: Not available
Six studies associating VDR ApaI gene polymorphism and urolithiasis risk were identified from the literature search. A total of 2074 cases and 2263 controls were included in our analysis. Between studies heterogeneity was detected in the allelic model and homozygous model but not in the dominant, recessive, over-dominant model and heterozygous model. In the pooled OR from overall studies, the ApaI variant showed no significant associations in any of the genetic models: allelic model (a vs. A: OR=1.04, 95% CI 0.89-1.23), dominant model (Aa+aa vs. AA: OR=1.06, 95% CI 0.89-1.26), recessive model (aa vs. AA+Aa: OR=1.14, 95% CI 1.00-1.30), over-dominant model (Aa vs. AA+aa: OR=0.91, 95% CI 0.81-1.03), homozygous model (aa vs. AA: OR=1.07, 95% CI 0.74-1.56), heterozygous model (Aa vs. AA: OR=1.00, 95% CI 0.83-1.21) or minor allele homozygote versus heterozygote model (aa vs. Aa: OR=1.14, 95% CI 0.99-1.30) (Tab. 2, Fig. 2). However, in the subgroup analysis, a significant association was found among the East Asian in the recessive model (aa vs. AA+ Aa: OR=1.17, 95% CI 1.00-1.30) and pairwise minor allele homozygote versus heterzygote model (aa vs. Aa: OR=1.16, 95% CI 1.01-1.34) but not among the Southwest Asian. Funnel plot and Egger’s test were used to detect publication bias. However, publication bias was not detected (p>0.05).
| ApaI Genetic contrasts | Subgroup | No. of studies | Q test P value |
Model selected | OR (95% CI) | p value | Egger’s p value |
| a vs. A | Overall | 6 | 0.04 | Random | 1.04 (0.89-1.23) | 0.60 | 0.24 |
| East Asian | 5 | 0.02 | Random | 1.05 (0.87-1.26) | 0.60 | 0.34 | |
| Southwest Asian | 1 | - | Fixed | 0.97 (0.69-1.37) | 0.86 | - | |
| aa vs AA+Aa | Overall | 6 | 0.07 | Random | 1.10 (0.88-1.38) | 0.40 | 0.41 |
| East Asian | 5 | 0.11 | Fixed | 1.17 (1.02-1.33) | 0.02 | 0.75 | |
| Southwest Asian | 1 | - | Fixed | 0.62 (0.29-1.30) | 0.21 | - | |
| Aa+aa vs. AA | Overall | 6 | 0.07 | Random | 1.04 (0.79-1.38) | 0.76 | 0.58 |
| East Asian | 5 | 0.04 | Random | 1.00 (0.72-1.41) | 0.96 | 0.59 | |
| Southwest Asian | 1 | - | Fixed | 1.17 (0.0.71-1.92) | 0.54 | - | |
| Aa vs. AA+aa | Overall | 6 | 0.35 | Fixed | 0.91 (0.81-1.03) | 0.14 | 0.79 |
| East Asian | 5 | 0.73 | Fixed | 0.88 (0.78-1.00) | 0.06 | 0.52 | |
| Southwest Asian | 1 | - | Fixed | 1.42 (0.88-2.28) | 0.15 | - | |
| aa vs. AA | Overall | 6 | 0.03 | Random | 1.07 (0.74-1.56) | 0.71 | 0.15 |
| East Asian | 5 | 0.03 | Random | 1.13 (0.76-1.70) | 0.54 | 0.28 | |
| Southwest Asian | 1 | - | Fixed | 0.72 (0.32-1.62) | 0.43 | - | |
| aa vs. Aa | Overall | 6 | 0.12 | Fixed | 1.14 (0.99-1.30) | 0.07 | 0.64 |
| East Asian | 5 | 0.26 | Fixed | 1.16 (1.01-1.34) | 0.03 | 0.83 | |
| Southwest Asian | 1 | - | Fixed | 0.55 (0.25-1.20) | 0.14 | - | |
| Aa vs. AA | Overall | 6 | 0.14 | Fixed | 1.00 (0.83-1.21) | 0.98 | 0.71 |
| East Asian | 5 | 0.13 | Fixed | 0.96 (0.78-1.18) | 0.71 | 0.62 | |
| Southwest Asian | 1 | - | Fixed | 1.31 (0.78-2.19) | 0.31 | - | |
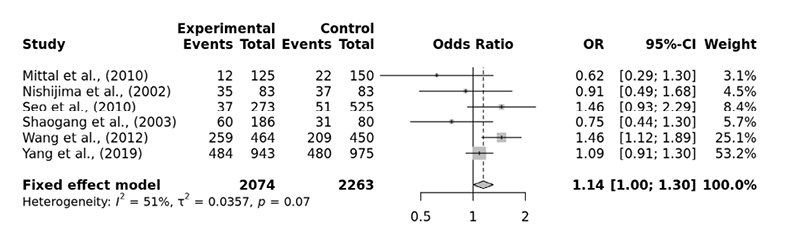
Fig. 2. Association analysis of ApaI variant with urolithiasisna
A total of 4 papers consisting of 959 cases and 870 controls were included to determine the correlation between BsmI and the risk of urolithiasis. Between studies heterogeneity was not detected. From the pooled OR in this meta-analysis we observed that the BsmI variant had no association with urolithiasis risk in the allelic comparison (b vs. B: OR=1.13, 95% CI 0.95-1.35), dominant model (Bb+bb vs. BB: OR=1.17, 95% CI 0.85-1.61), recessive model (bb vs. BB+Bb: OR=1.16, 95% CI 0.91-1.47), over-dominant model (Bb vs. BB+bb: OR=0.95, 95% CI 0.75-1.19), homozygous model (bb vs. BB: OR=1.23, 95% CI 0.83-1.81), minor allele homozygote versus heterzygote model (bb vs. Bb: OR=1.13, 95% CI 0.88-1.45) or heterozygous model (Bb vs. BB: OR=1.12, 95% CI 0.79-1.60) (Tab. 3). Moreover, subgroup analysis also rejected any significant association between VDR BsmI variant with urolithiasis under any genetic model in East and Southwest Asian. Funnel plot and Egger’s test were performed to estimate the publication bias, and no publication bias was observed (p>0.05).
| BsmI Genetic contrasts | Subroup | No. of studies | Q test P value |
Model selected | OR (95% CI) | p value | Egger’s p value |
| b vs. B | Overall | 4 | 0.66 | Fixed | 1.13 (0.95-1.35) | 1.16 | 0.60 |
| East Asian | 2 | 0.60 | Fixed | 1.02 (0.75-1.39) | 0.88 | - | |
| Southwest Asian | 2 | 0.41 | Fixed | 1.19 (0.96-1.47) | 0.11 | - | |
| bb vs. BB+Bb | Overall | 4 | 0.86 | Fixed | 1.16 (0.91-1.47) | 0.23 | 0.63 |
| East Asian | 2 | 0.88 | Fixed | 1.08 (0.76-1.54) | 0.65 | - | |
| Southwest Asian | 2 | 0.48 | Fixed | 1.23 (0.88-1.70) | 0.22 | - | |
| Bb+bb vs. BB | Overall | 4 | 0.22 | Fixed | 1.17 (0.85-1.61) | 0.33 | 0.62 |
| East Asian | 2 | 0.85 | Fixed | 0.80 (0.38-1.69) | 0.56 | - | |
| Southwest Asian | 2 | 0.07 | Random | 1.30 (0.69-2.44) | 0.41 | - | |
| Bb vs. BB+bb | Overall | 4 | 0.07 | Random | 0.99 (0.68-1.45) | 0.96 | 0.69 |
| East Asian | 2 | 0.80 | Fixed | 0.89 (0.63-1.24) | 0.48 | - | |
| Southwest Asian | 2 | 0.01 | Random | 1.14 (0.49-2.68) | 0.76 | - | |
| bb vs. BB | Overall | 4 | 0.84 | Fixed | 1.23 (0.83-1.81) | 0.31 | 0.30 |
| East Asian | 2 | 0.80 | Fixed | 0.83 (0.26-2.66) | 0.76 | - | |
| Southwest Asian | 2 | 0.58 | Fixed | 1.29 (0.85-1.94) | 0.23 | - | |
| bb vs. Bb | Overall | 4 | 0.39 | Fixed | 1.13 (0.88-1.45) | 0.35 | 0.69 |
| East Asian | 2 | 0.93 | Fixed | 1.11 (0.78-1.59) | 0.56 | - | |
| Southwest Asian | 2 | 0.08 | Random | 1.03 (0.53-2.04) | 0.92 | - | |
| Bb vs. BB | Overall | 4 | 0.09 | Random | 1.09 (0.61-1.97) | 0.77 | 0.77 |
| East Asian | 2 | 0.83 | Fixed | 0.75 (0.32-1.74) | 0.50 | - | |
| Southwest Asian | 2 | 0.02 | Random | 1.31 (0.53-3.27) | 0.56 | - | |
For the association between FokI polymorphism of VDR gene and the risk of urolithiasis, a total of 11 papers consisting of 2143 cases and 2454 controls were included in our analysis. Heterogeneity between the studies was detected in the allelic, dominant, over-dominant and heterozygous models. The ff genotype of the FokI recessive model presented a significantly higher risk of urolithiasis by 1.27 fold (ff vs. FF+Ff: OR=1.27, 95% CI 1.07-1.51), by 1.24 fold in the homozygous model (ff vs. FF: OR=1.24, 95% CI 1.02-1.52) and by 1.26 fold in the minor allele homozygote versus heterzygote model (ff vs. Ff: OR=1.26, 95% CI 1.04-1.51) (Tab. 4, Fig. 3, 4 and 5). However, the allelic (f vs. F: OR= 1.06, 95% CI 0.88-1.28), dominant (Ff+ff vs. FF: OR= 1.01, 95% CI 0.72-1.40), over-dominant (Ff vs. FF+ff: OR= 0.88, 95% CI 0.64-1.22) and heterozygous model (Ff vs. FF: OR= 0.96, 95% CI 0.68-1.35) showed no significant association. In the subgroup analysis ff genotype among the East Asian are found to have 1.32 fold significantly higher urolithiasis risk than Ff genotype (OR=1.32, 95% CI 1.08-1.60). Egger’s test was performed to estimate the publication bias. Publication bias was not observed (p>0.05).
| FokI Genetic contrasts | Subgroup | No. of studies | Q test P value |
Model selected | OR (95% CI) | p value | Egger’s p value |
| f vs. F | Overall | 11 | 0.00 | Random | 1.06 (0.88-1.28) | 0.51 | 0.54 |
| East Asian | 6 | 0.01 | Random | 1.13 (0.92-1.38) | 0.24 | 0.82 | |
| Southwest Asian | 5 | 0.00 | Random | 0.96 (0.65-1.41) | 0.83 | 0.33 | |
| ff vs. FF+Ff | Overall | 11 | 0.11 | Fixed | 1.27 (1.07-1.51) | 0.01 | 0.52 |
| East Asian | 6 | 0.06 | Random | 1.32 (0.99-1.77) | 0.06 | 0.91 | |
| Southwest Asian | 5 | 0.55 | Fixed | 0.90 (0.52-1.55) | 0.70 | 0.65 | |
| Ff+ff vs. FF | Overall | 11 | 0.00 | Random | 1.01 (0.72-1.40) | 0.97 | 0.54 |
| East Asian | 6 | 0.06 | Random | 1.06 (0.82-1.36) | 0.66 | 0.73 | |
| Southwest Asian | 5 | 0.00 | Random | 0.88 (0.40-1.93) | 0.74 | 0.38 | |
| Ff vs. FF+ff | Overall | 11 | 0.00 | Random | 0.88 (0.64-1.22) | 0.45 | 0.48 |
| East Asian | 6 | 0.49 | Fixed | 0.87 (0.76-1.01) | 0.07 | 0.46 | |
| Southwest Asian | 5 | 0.00 | Random | 0.88 (0.38-2.03) | 0.77 | 0.36 | |
| ff vs. FF | Overall | 11 | 0.15 | Fixed | 1.24 (1.02-1.52) | 0.03 | 0.50 |
| East Asian | 6 | 0.02 | Random | 1.29 (0.89-1.88) | 0.18 | 0.97 | |
| Southwest Asian | 5 | 0.97 | Fixed | 0.94 (0.54-1.65) | 0.84 | 0.68 | |
| ff vs. Ff | Overall | 11 | 0.10 | Fixed | 1.26 (1.04-1.51) | 0.02 | 0.59 |
| East Asian | 6 | 0.21 | Fixed | 1.32 (1.08-1.60) | 0.01 | 0.94 | |
| Southwest Asian | 5 | 0.17 | Fixed | 0.83 (0.47-1.46) | 0.53 | 0.53 | |
| Ff vs. FF | Overall | 11 | 0.00 | Random | 0.96 (0.68-1.35) | 0.80 | 0.47 |
| East Asian | 6 | 0.28 | Fixed | 0.98 (0.84-1.16) | 0.79 | 0.90 | |
| Southwest Asian | 5 | 0.00 | Random | 0.88 (0.38-2.02) | 0.76 | 0.36 | |
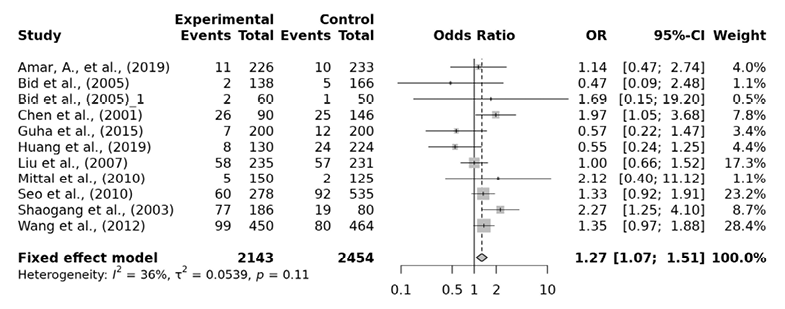
Fig. 3. FokI ff vs. FF+Ff forest plot
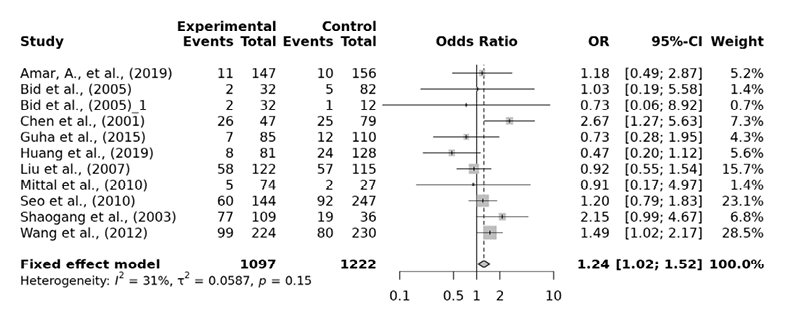
Fig. 4. FokI ff vs. FF forest plot
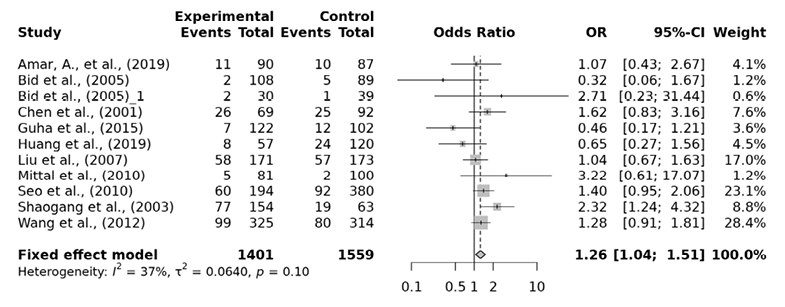
Fig. 5. FokI ff vs. Ff forest plot
Ten studies associating VDR TaqI gene polymorphism and urolithiasis risk were included in our analysis comprising of a total of 2811 cases and 2920 controls. Between studies heterogeneity were detected in the dominant, overdominant and heterogyzous model. The pooled OR from this meta-analysis showed no significant association of the TaqI variant with urolithiasis risk in the allelic comparison (t vs. T: OR=1.09, 95% CI 0.97-1.23), dominant model (Tt+tt vs. TT: OR=1.09, 95% CI 0.86 -1.37), recessive model (tt vs. TT+Tt: OR=1.47, 95% CI 1.09-2.00), over-dominant model (Tt vs. TT+tt: OR=0.97, 95% CI 0.78-1.20), homozygous model (tt vs. TT: OR=1.48, 95% CI 1.07-2.04), pairwise minor allele homozygote versus heterozygote model (tt vs. Tt: OR=1.47, 95% CI 1.07-2.03) or heterozygous model (Tab. 5: Tt vs. TT: OR=1.03, 95% CI 0.81-1.31).
| TaqI Genetic contrasts | Subgroup | No. of studies | Q test >P value |
Model selected | OR (95% CI) | p value | Egger’s p value |
| t vs. T | Overall | 10 | 0.08 | Random | 1.09 (0.93-1.26) | 0.29 | 0.05 |
| East Asian | 5 | 0.36 | Fixed | 1.08 (0.91-1.32) | 0.38 | 0.11 | |
| Southwest Asian | 5 | 0.03 | Random | 1.14 (0.83-1.55) | 0.56 | 0.25 | |
| tt vs. TT+Tt | Overall | 10 | 0.20 | Fixed | 1.47 (1.09-2.00) | 0.34 | 0.29 |
| East Asian | 5 | 0.74 | Fixed | 1.12 (0.63-2.01) | 0.70 | 0.93 | |
| Southwest Asian | 5 | 0.04 | Random | 1.63 (1.14-2.34) | 0.25 | 0.02 | |
| Tt+tt vs. TT | Overall | 10 | 0.03 | Random | 1.09 (0.86-1.37) | 0.40 | 0.06 |
| East Asian | 5 | 0.38 | Fixed | 1.09 (0.89- 1.33) | 0.40 | 0.03 | |
| Southwest Asian | 5 | 0.01 | Random | 1.03 (0.64-1.67) | 0.78 | 0.34 | |
| Tt vs. TT+tt | Overall | 10 | 0.01 | Random | 0.97 (0.78-1.20) | 0.76 | 0.33 |
| East Asian | 5 | 0.61 | Fixed | 1.07 (0.88-1.31) | 0.49 | 0.01 | |
| Southwest Asian | 5 | 0.00 | Random | 0.81 (0.54-1.22) | 0.84 | 0.82 | |
| tt vs. TT | Overall | 10 | 0.44 | Fixed | 1.48 (1.07-2.04) | 0.09 | 0.51 |
| East Asia | 5 | 0.66 | Fixed | 1.24 (0.67-2.28) | 0.49 | 0.96 | |
| Southwest Asia | 5 | 0.16 | Fixed | 1.58 (1.08-2.31) | 0.12 | 0.10 | |
| tt vs. Tt | Overall | 10 | 0.07 | Random | 1.19 (0.80-1.78) | 0.39 | 0.42 |
| East Asia | 5 | 0.83 | Fixed | 1.00 (0.54-1.83) | 0.99 | 0.93 | |
| Southwest Asia | 5 | 0.01 | Random | 1.71 (1.17-2.51) | 0.35 | 0.06 | |
| Tt vs. TT | Overall | 10 | 0.01 | Random | 1.03 (0.81-1.31) | 0.52 | 0.12 |
| East Asian | 5 | 0.49 | Fixed | 1.09 (0.89-1.33) | 0.43 | 0.01 | |
| Southwest Asian | 5 | 0.00 | Random | 0.91 (0.55-1.51 | 0.92 | 0.36 | |
Additionally, the subgroup analysis also indicated no signgificant association of urolithiasis risk with TaqI variant among the East and Southwest Asian subgroup. Publication bias (p<0.05) was detected among the East Asian subgroup in the dominant model, over-dominant model and heterozygous model and in the recessive model among the Southwest Asian subgroup.
Several sensitivity tests were performed to evaluate the robustness of our findings by sequentially removing one study in turn. The analysis is repeated excluding one study each time in order to visualize if any study has a significantly greater contribution to overall statistics than the other studies. However, the pooled ORs of the significantly associated genetic models were not substantially altered and was within the 95% CI, indicating that no single study influenced the stability of the results obtained in this meta-analysis (Fig. 6, 7, 8, and 9). Egger’s test with p value of <0.05 defined as having potential publication bias was used to evaluate potential publication bias. However, in this meta-analysis, except for TaqI polymorphism, no publication bias was observed both in overall and subgroup analyses. The funnel plots of the significantly associated genetic models also shows to be symmetrical (Fig. 10, 11, 12 and 13).
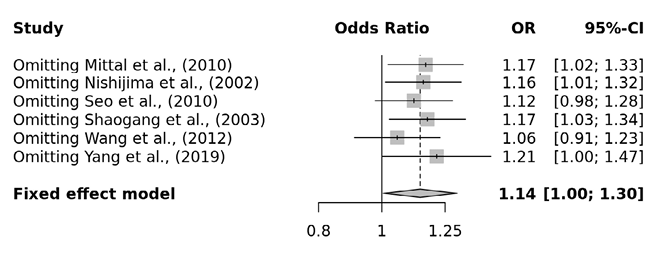
Fig. 6. Apa aa vs. AA+Aa sensitivity plot
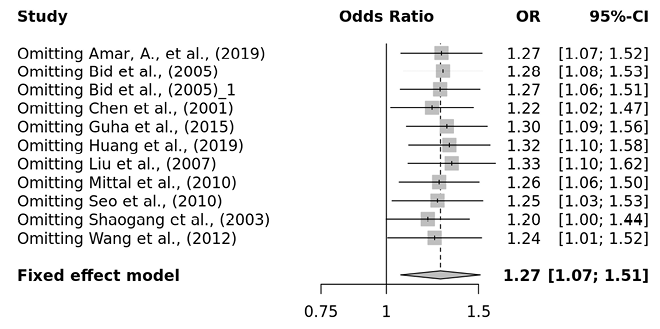
Fig. 7. FokI ff vs. FF+Ff sensitivity plot
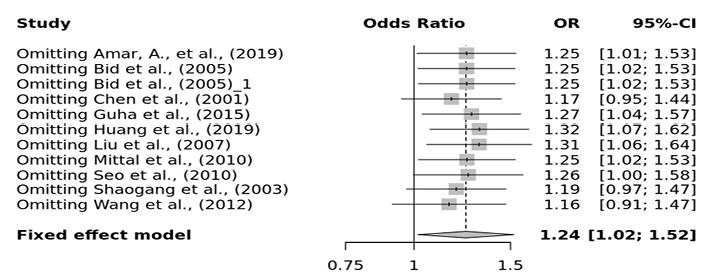
Fig. 8. FokI ff vs. FF sensitivity plot
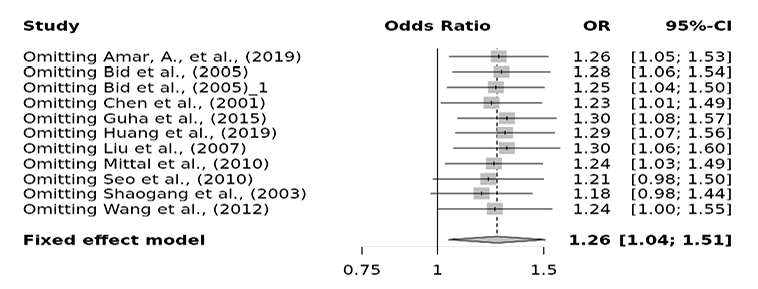
Fig. 9. FokI ff vs. Ff sensitivity plot
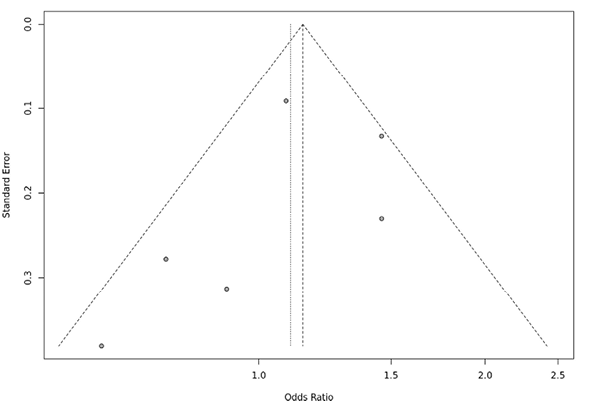
Fig. 10. ApaI aa vs. AA+Aa funnel plot
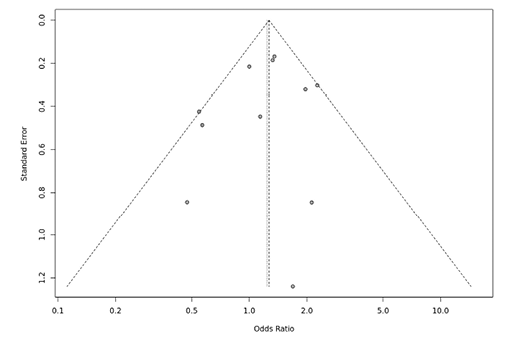
Fig. 11. FokI ff vs. FF+Ff funnel plot
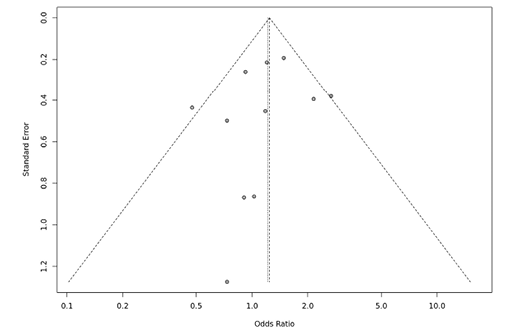
Fig. 12. FokI ff vs. FF funnel plot
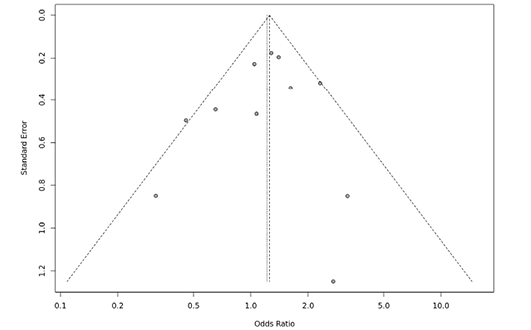
Fig. 13. FokI ff vs.Ff funnel plot
In this meta-analysis, a total of 17 articles reporting the relationship between VDR ApaI, BsmI, FokI, and TaqI gene variants and urolithiasis susceptibility in the Asian population were analyzed. In addition to the earlier allelic, recessive, dominant, over-dominant, pair-wise homozygous and heterozygous genetic models, one new pair-wise genetic model, which compares minor allele homozygote to the heterozygote (aa vs. Aa; bb vs. Bb; ff vs. Ff; tt vs. Tt), has been included in this meta-analysis. The results of the pooled analysis revealed that VDR gene polymorphism was significantly associated with urolithiasis risk in the Asian population. Six studies were included for evaluating the association of ApaI variant, with urolithiasis risk, and was found that ApaI aa genotype had a 1.17 fold increased risk of urolithiasis in the recessive model (aa vs. AA+Aa: OR=1.17, 95% CI 1.02-1.33) and 1.16 fold increased risk in the pair-wise aa vs. Aa model (OR=1.16, 95% CI 1.01-1.34) among the East Asian population but not among the Southwest Asian. Similar findings of positive significant association of ApaI polymorphism with urolithiasis risk in the East-Asians has also been reported by Imani et al. (2020), where the ‘a’ allele showed a significant association with a higher urolithiasis risk compared to A allele. According to their result, the aa genotype in recessive model had a higher risk of urolithiasis compared to AA and Aa genotypes (OR=1.20, 95% CI 1.05-1.37, p<0.001), a allele in allelic model had 1.15 fold increased risk of urolithiasis (OR=1.15, 95% CI 1.05-1.26, p<0.001) and aa genotype in the homozygous mode was at 1.40 fold higher risk of urolithiasis than AA genotype (OR=1.40, 95% CI 1.12-1.75, p <0.001). However, our finding is in contrast with Zhang et al. (2013), in which the AA or Aa genotypes of ApaI variant to have been reported to have an increased risk compare to aa genotype. This contrasting finding could have arised due to difference in number of studies included as we had language restricition and included only those studies available in English.
The outcome of this meta-analysis showed no significant association of BsmI polymorphism with urolithiasis risk across all the seven genetic models: allelic (OR=1.13, 95% CI 0.95-1.35), dominant model (OR=1.17, 95% CI 0.85-1.61), recessive model (OR=1.16, 95% CI 0.0.91-1.47), over-dominant model (OR=0.95, 95% CI 0.75-1.19), homozygous model (OR=1.23, 95% CI 0.83-1.81), minor allele homozygote versus heterozygote model (OR=1.13, 95% CI 0.88-1.45) or heterozygous model (OR=1.12, 95% CI 0.79-1.60). The subgroup analysis also did not show any association of BsmI variant with urolithiasis in the East Asian and the Southwest Asian subgroup. However, the results are less robust since there is an inadequate number of a primary study included in this meta-analysis. Nonetheless, the insignificant association between BsmI variant and urolithiasis risk are still consistent with other meta-analysis (Chen et al. 2020; Daryanto et al. 2020; González-Castro et al. 2019; Imani et al. 2020; Lin et al. 2011; Zhang et al. 2013). According to the literature, the minor f allele of the FokI variant introduces a start codon leading to a three amino acid longer VDR protein (Gross et al. 1998),which in turn influences the VDR protein activity and results in less effective transcriptional activator (Cakir et al. 2016; Dastani et al. 2013). However, from our analysis, the overall finding of the FokI variant reported ff genotype in the recessive model presents a higher risk of urolithiasis by 1.27 fold (ff vs. FF+Ff: OR=1.27, 95% CI 1.07-1.51), by 1.24 fold in the homozygous model (ff vs. FF OR=1.24, 95% CI 1.02-1.52) and by 1.26 fold in the minor allele homozygote versus the heterozygote model (ff vs. Ff: OR=1.26, 95% CI 1.04-1.51) suggesting a protective association of F over f allele. Moreover, subgroup analysis also revealed ff genotype to be at 1.32 fold increased risk of urolithiasis than Ff genotype (ff vs. Ff: OR=1.32, 95% CI 1.08-1.60) among the east Asian population. Previous meta-analysis had also reported the ff+Ff genotype in the dominant genetic model and f allele presented with a marginally positive relationship to susceptibility for urolithiasis in the Asian subgroup (Lin et al. 2011). On the other hand, Zhou et al. (2015) found FF genotype is associated with increased urine calcium levels in urolithiasis but not with ff or Ff genotypes. However, association of FokI variant with urolithiasis risk among Asians has been conflicting as number of studies rejected any significant association (Chen et al. 2020; Imani et al. 2020; Yang et al. 2019; Zhang et al. 2013).
As per literature, the TaqI variant does not modify the VDR protein structure but can influence the translation efficiency and/or stability of the RNA, which might in turn affect the development of urolithiasis (Jurutka et al. 2001; Uitterlinden et al. 2004). Furthermore, it has also been found that individuals with tt genotype had significantly higher VDR mRNA levels compared to those with TT genotype (Carling et al. 1998), which suggests that the t allele has an increased susceptibility. However, from our analysis, the TaqI polymorphism was not found to be associated with urolithiasis risk among the Asian population under any of the genetic models considered. Further, subgroup analysis also did not result in any significant associations. However, earlier meta-analysis by Yang et al. (2019) reported that TaqI polymorphism of the VDR gene is associated with urolithiasis risk in theAsian population. Their result indicated the TT genotype has a protective association with urolithiasis risk while the heterozygote Tt genotype has a significantly higher risk of urolithiasis over the homozygotes. Similar findings by Zhang et al. (2013) reported the tt or Tt genotypes with 1.39 fold increases the risk of urolithiasis compared to the TT genotype. Also, according to Chen et al. (2020), significant urolithiasis risk with TaqI variant was identified among Asians where the Tt genotype had 1.33 fold increased risk and TT genotype had a protective association with urolithiasis. Nonetheless, our analysis did not result in any significant association of TaqI variant with urolithiasis risk.
In summary, findings from our meta-analysis supports the fact that some of the VDR gene polymorphisms are associated with an increase in probability of urolithiasis among the Asian population under certain genetic models. Our results indicated that VDR ApaI and FokI polymorphisms are associated with risk of urolithiasis. The results from our pooled analysis indicated ApaI aa genotype are associated with urolithiasis compared to AA or Aa genotypes. Additionally, the minor f allele of FokI variant was indicated to be the risk allele in susceptibility to urolithiasis while F allele to be protective. Moreover, from the subgroup analysis, the ff genotype of FokI and aa genotype of ApaI were associated with higher risk of urolithiasis among the East Asian while the same was not the case among the Southwest Asian.
Efforts have been made to test the publication bias and conducting sensitivity analysis to acquire a more accurate estimate based off the available studies. However, there were heterogeneities in some of the included studies and it may affect the accuracy of the analysis. Many more detailed and well-designed case-control studies needs to be carried out to validate the role of VDR gene polymorphisms in susceptibility to urolithiasis. This review highlights the varied association of VDR gene polymorphism with urolithiasis among the Asian population, which provides the importance of population stratification in designing future association studies among the Asian population with respect to VDR gene polymorphisms or any other genetic trait.
Authors’ contribution
Sanjenbam Yaiphaba (SY) originated the idea/concept of the study. Malvika Yumnam (MY) and SY acquired and organised the data. MY performed statistical analysis, interpreted data, and drafted the manuscript. SY revised the manuscript. Both authors read and approved the final version of the manuscript.
Funding source
This study is part of a project funded by DST-SERB under Core Research Grant (CRG/2020/004697).
Declaration of competing interest
The authors declare no conflict of interest related to this publication.
Amar A, Afzal A, Hussain SA, Hameed A, Khan AR, Shakoor M, et al. 2020. Association of Vitamin D Receptor Gene Polymorphisms and Risk of Urolithiasis: Results of a Genetic Epidemiology Study and Comprehensive Meta-Analysis. Urolithiasis 48 (5): 385–401. https://doi.org/10.1007/s00240-019-01157-7
Arababadi MK, Abousaidi H, Hassanshahi G, Pourfathollah AA, Daneshmandi S, Akbarpour V, et al. 2011. Polymorphisms within Exon 9, But Not Intron 8, of the Vitamin D Receptor Gene Are Associated with Asthma. Iranian Journal of Basic Medical Sciences 14 (3): 225–230.
Arai H, Miyamoto K, Taketani Y, Yamamoto H, Iemori Y, Morita K, et al. 1997. A Vitamin D Receptor Gene Polymorphism in the Translation Initiation Codon: Effect on Protein Activity and Relation to Bone Mineral Density in Japanese Women. Journal of Bone and Mineral Research 12 (6): 915–921.
Bid HK, Chaudhary H, Mittal RD. 2005a. Association of Vitamin-D and Calcitonin Receptor Gene Polymorphism in Paediatric Nephrolithiasis. Pediatric Nephrology 20 (6): 773–776. https://doi.org/10.1007/s00467-005-1846-4
Bid HK, Kumar A, Kapoor R, Mittal RD. 2005b. Association of Vitamin D Receptor-Gene (FokI) Polymorphism with Calcium Oxalate Nephrolithiasis. Journal of Endourology 19 (1): 111–115.
Cakir OO, Yilmaz A, Demir E, Incekara K, Kose MO, Tunali NE. 2016. Association of the BsmI, ApaI, TaqI, Tru9I and FokI Polymorphisms of the Vitamin D Receptor Gene with Nephrolithiasis in the Turkish Popula-Tion. Endourology and Stone Disease 13 (1): 2509–2518.
Carling T, Rastad J, Åkerström G, Westin G. 1998. Vitamin D Receptor (VDR) and Parathyroid Hormone Messenger Ribonucleic Acid Levels Correspond to Polymorphic VDR Alleles in Human Parathyroid Tumors. Journal of Clinical Endocrinology and Metabolism 83 (7): 2255–2259. https://academic.oup.com/jcem/article/83/7/2255/2865167
Chen G, Hu C, Song Y, Xiu M, Liang W, Ou N, et al. 2020. Relationship between the Apai (Rs7975232), Bsmi (Rs1544410), Foki (Rs2228570), and Taqi (Rs731236) Variants in the Vitamin D Receptor Gene and Urolithiasis Susceptibility: An Updated Meta-Analysis and Trial Sequential Analysis. Frontiers in Genetics 11. https://doi.org/10.3389/fgene.2020.00234
Chen W.-C, Chen H.-Y, Lu H.-F, Hsu C.-D, Tsai F.-J. 2001a. Association of the Vitamin D Receptor Gene Start Codon Fok I Polymorphism with Calcium Oxalate Stone Disease. BJU International 87:168–171.
Chen W-C, Chen H-Y, Hsu C-D, Wu J-Y, Tsai F-J. 2001b. No Association of Vitamin D Receptor Gene BsmI Polymorphisms with Calcium Oxalate Stone Formation. Molecular Urology 5 (1): 7–10.
Daryanto B, Purnomo BB, Gunawan A, Mayasari ED, Kusumaningrum AG, Tamara F, et al. 2020. The Association between Vitamin D Receptor Gene Polymorphisms and the Risk of Nephrolithiasis: A Meta-Analysis. Meta Gene 23 (February). https://doi.org/10.1016/j.mgene.2019.100628
Dastani Z, Li R, Richards B. 2013. Genetic Regulation of Vitamin D Levels. Calcified Tissue International 92 (2): 106–117. https://doi.org/10.1007/s00223-012-9660-z
González-Castro TB, Blachman-Braun R, Hernández-Díaz Y, Tovilla-Zárate CA, Pérez-Hernández N, Moscardi PRM, et al. 2019a. Association of Vitamin D Receptor Polymorphisms and Nephrolithiasis: A Meta-Analysis. Gene 711 (August):143936. https://doi.org/10.1016/j.gene.2019.06.026
Gross C, Krishnan AV, Malloy PJ, Eccleshall TR, Zhao X-Y, Feldman D. 1998. The Vitamin D Receptor Gene Start Codon Polymorphism: A Functional Analysis of FokI Variants. Journal of Bone and Mineral Research 13 (11): 1691–1699.
Guha M, Bankura B, Ghosh S, Pattanayak AK, Ghosh S, Pal DK, et al. 2015. Polymorphisms in CaSR and CLDN14 Genes Associated with Increased Risk of Kidney Stone Disease in Patients from the Eastern Part of India. PLoS ONE 10 (6): e0130790. https://doi.org/10.1371/journal.pone.0130790
Fallahzadeh MH, Zare J, Al-Hashemi GH, Derakhshan A, Basiratnia M, Arasteh MA, et al. 2012. Elevated Serum Levels of Vitamin D in Infants With Urolithiasis. Iranian Journal of Kidney Diseases 6:186–191. www.ijkd.org
Huang Y, Peng Q, Bao M, Liu C, Wu K, Zhou S. 2019. Biochemical Metabolic Levels and Vitamin D Receptor FokⅠ Gene Polymorphisms in Uyghur Children with Urolithiasis. PLoS ONE 14 (2). https://doi.org/10.1371/journal.pone.0212183
Imani D , Razi R, Khosrojerdi A, Lorian K, Motallebnezhad M, Rezaei R, et al. 2020a. Vitamin D Receptor Gene Polymorphisms and Susceptibility to Urolithiasis: A Meta-Regression and Meta-Analysis. BMC Nephrology 21 (1): 263. https://doi.org/10.1186/s12882-020-01919-1
Jawad ZN, Awad W. 2020a. Association of Urokinase and Vitamin D Receptor Genes SNPs and Urolithiasis in an Iraqi Population. Meta Gene 24 (June). https://doi.org/10.1016/j.mgene.2020.100679
Jurutka PW, Whitfield GK, Hsieh J-C, Thompson PD, Haussler CA, Haussler MR. 2001. Molecular Nature of the Vitamin D Receptor and Its Role in Regulation of Gene Expression. Reviews in Endocrine & Metabolic Disorders 2:203–216.
Labuda M, Fujiwara TM, Ross MV, Glorieux FH, Morgan K, Garcia-heras J, et al. 1992. Two Hereditary Defects Related to Vitamin D Metabolism Map to the Same Region of Human Chromosome 12q13-14. Journal of Bone and Mineral Research 7 (12): 1447–1453.
Lee Y-H, Huang W-C, Tsai J-Y, Lu C-M, Chen W-C, Lee M-H, et al. 2002. Epidemiological Studies on the Prevalence of Upper Urinary Calculi in Taiwan. Urolgia Internationalis 68:172–177. www.karger.com/journals/uin
Lin Y, Mao Q, Zheng X, Chen H, Yang K, Xie L. 2011. Vitamin D Receptor Genetic Polymorphisms and the Risk of Urolithiasis: A Meta-Analysis. Urologia Internationalis 86 (3): 249–255. https://doi.org/10.1159/000323949
Liu CC, Huang CH, Wu WJ, Huang SP, Chou YH, Li CC, et al. 2007. Association of Vitamin D Receptor (Fok-I) Polymorphism with the Clinical Presentation of Calcium Urolithiasis. BJU International 99 (6): 1534–1538.
Luchini C, Stubbs B, Solmi M, Veronese N. 2017. Assessing the quality of studies in meta-analyses: Advantages and limitations of the Newcastle Ottawa Scale. World J Meta-Anal 5(4):80:84.
Martorell-Marugan J, Toro-Dominguez D, Alarcon-Riquelme ME, Carmona-Saez P. 2017. MetaGenyo: A Web Tool for Meta-Analysis of Genetic Association Studies. BMC Bioinformatics 18 (1). https://doi.org/10.1186/s12859-017-1990-4
Mittal R D, Mishra D K, Srivastava P, Manchanda P, Bid H K, Kapoor R, et al. 2010. Polymorphisms in the Vitamin D Receptor and the Androgen Receptor Gene Associated with the Risk of Urolithiasis. Indian Journal of Clinical Biochemistry 25 (2): 119–126.
Miyamoto K-I, Kesterson RA, Yamamoto H, Taketani Y, Nishiwaki E, Tatsumi S, et al. 1997. Structural Organization of the Human Vitamin D Receptor Chromosomal Gene and Its Promoter. Molecular Endocrinology 11 (8): 1165–1179.
Morrison NA, Oi JC, Tokita A, Kelly PJ, Crofts L, Nguyen TV, et al. 1994. Prediction of Bone Density from Vitamin D Receptor Alleles. Nature 367:284–287.
Nishijima S, Sugaya K, Naito A, Morozumi M, Hatano T, Ogawa Y. 2002. Association of Vitamin D Receptor Gene Polymorphism with Urolithiasis. The Journal of Urology 167:2188–91.
Ou C, Zhao HL, Zhu B, Huang LS, Li PZ, Lao M. 2014. Association of Vitamin D Receptor Gene Polymorphism with the Risk of Renal Cell Carcinoma: A Meta-Analysis. Journal of Receptors and Signal Transduction 34 (6): 463–468. https://doi.org/10.3109/10799893.2014.919593
Parvaresh S, Kordestani Z, Maftuhi M, Mohammadi M, Miri Karam Z, Salari Nasab S. 2022. Association of Vitamin D Receptor TaqI Gene Polymorphisms and Susceptibility to Pediatric Urolithiasis in the Iranian Population. Gene, Cell and Tissue 9 (4): e121997. https://doi.org/10.5812/gct-121997
Relan V, Khullar M, Singh SK, Sharma SK. 2004. Association of Vitamin d Receptor Genotypes with Calcium Excretion in Nephrolithiatic Subjects in Northern India. Urological Research 32 (3): 236–240. https://doi.org/10.1007/s00240-004-0414-x
Seo IY, Kang IH, Chae SC, Park SC, Lee YJ, Yang YS, et al. 2010. Vitamin D Receptor Gene Alw I, Fok I, Apa I, and Taq I Polymorphisms in Patients With Urinary Stone. Urology 75 (4): 923–927. https://doi.org/10.1016/j.urology.2009.10.006
Shaogang W, Jihong L, Shaoqun L, Zhangqun Y. 2003. Association of Vitamin D Receptor Gene Polymorphisms with Calcium Oxalate Calculus Disease. Journal of Huazhong University of Science and Technology (Med Sci)’ 23 (1): 38–41.
Subaşı B, Gökçe I, Delil K, Alpay H. 2017. Vitamin D Receptor Gene Polymorphisms in Children with Kidney Stone Disease. Turkish Journal of Pediatrics 59 (4): 404–409. https://doi.org/10.24953/turkjped.2017.04.006
Uitterlinden AG, Fang Y, Van Meurs JBJ, Pols HAP, Van Leeuwen JP.T.M. 2004. Genetics and Biology of Vitamin D Receptor Polymorphisms. Gene 338 (2): 143–156. https://doi.org/10.1016/j.gene.2004.05.014
Wang S, Wang X, Wu J, Lin Y, Chen H, Zheng X, et al. 2012. Association of Vitamin D Receptor Gene Polymorphism and Calcium Urolithiasis in the Chinese Han Population. Urological Research 40 (4): 277–284. https://doi.org/10.1007/s00240-011-0438-y
Yang SK, Song N, Wang YY, Li AM, Liu J, Deng F, et al. 2019. Association of Vitamin D Receptor Gene Polymorphism With the Risk of Nephrolithiasis. Therapeutic Apheresis and Dialysis 23 (5): 425–436. https://doi.org/10.1111/1744-9987.12797
Yang Z, Wang Q, Zhong JF, Longkun L. 2019. Polymorphisms of the VDR Gene in Patients with Nephrolithiasis in a Han Chinese Population. Urolithiasis 47:149–154.
Zhang P, Nie W, Jiang H. 2013. Effects of Vitamin D Receptor Polymorphisms on Urolithiasis Risk: A Meta-Analysis. BMC Medical Genetics 14 (1). https://doi.org/10.1186/1471-2350-14-104
Zhou TB, Jiang ZP, Huang MF, Zhang R. 2015. Association of Vitamin D Receptor Gene Polymorphism with the Urine Calcium Level in Nephrolithiasis Patients. Journal of Receptors and Signal Transduction 35 (2): 127–132. https://doi.org/10.3109/10799893.2014.936462

