Available online at: https://doi.org/10.18778/1898-6773.86.4.08
 https://orcid.org/0000-0002-7826-8715
https://orcid.org/0000-0002-7826-8715
University of Lodz, Faculty of Biology and Environmental Protection, Department of Anthropology, 90-237, Poland
 https://orcid.org/0000-0001-9586-5389
https://orcid.org/0000-0001-9586-5389
University of Lodz, Faculty of Biology and Environmental Protection, Department of Anthropology, 90-237, Poland
 https://orcid.org/0000-0003-1001-7319
https://orcid.org/0000-0003-1001-7319
University of Lodz, Faculty of Biology and Environmental Protection, Department of Anthropology, 90-237, Poland
Biological Anthropology and Comparative Anatomy Research Unit, School of Medicine, University of Adelaide, South Australia 5005, Australia
 https://orcid.org/0000-0003-1523-1912
https://orcid.org/0000-0003-1523-1912
University of Lodz, Faculty of Biology and Environmental Protection, Department of Anthropology, 90-237, Poland
 https://orcid.org/0000-0002-0285-9092
https://orcid.org/0000-0002-0285-9092
Medical University of Lodz, Department of Sleep Medicine and Metabolic Disorders, Lodz, Poland
 https://orcid.org/0000-0001-6187-0833
https://orcid.org/0000-0001-6187-0833
Medical University of Lodz, Department of Sleep Medicine and Metabolic Disorders, Lodz, Poland
 https://orcid.org/0000-0002-6802-1997
https://orcid.org/0000-0002-6802-1997
University of Lodz, Faculty of Biology and Environmental Protection, Department of Anthropology, 90-237, Poland
ABSTRACT: In this study a widely debated association between 2D:4D digit ratio and age was investigated. The study material included 960 individuals (530 females and 430 males) from Central Poland aged between 6–79 years. The information about age was obtained via survey filled in by study participants or, if underaged, their parents. The direct measurements of the second and fourth finger were performed to assess the 2D:4D digit ratio. The 2D:4D digit ratios for the left hand were significantly correlated with age both among females and males. In women the 2D:4D digit ratios for the right hand were significantly correlated with age. There were also significant differences in digit ratio between age groups. The results of our study suggest that there might be an association between digit ratio and age, and the direction of the correlation might be related to the phase of the ontogenesis.
KEY WORDS: digit ratio, prenatal testosterone, prenatal sex hormones proportion.
The second to fourth finger ratio (2D:4D) is a proportion known since the second half of the nineteenth century. It has been shown that the prenatal exposure to sex steroids affects the 2D:4D ratio. For instance, higher exposure to androgens affects longer fourth finger and in opposite more estrogens affect shorter fourth finger resulting in typically man (2D:4D<1) and woman (2D:4D≥1) proportion (Lutchmaya et al. 2004). However, it is not certain to what extent the 2D:4D ratio precisely indicates the current sex steroids proportion (Manning et al. 2014). Some studies suggest that prenatal sex steroid exposure is associated with neither the level of circulating androgens nor estrogens (Muller et al. 2011; Hönekopp et al. 2007; Kowal et al. 2020).
Numerous studies have reported that 2D:4D digit ratio does not change during human ontogenesis (Manning et al. 2014; de Sanctis V et al. 2017). Moreover, there are studies indicating that estrogen and androgens levels during postnatal period are not related to 2D:4D, and thus do not affect 2D:4D digit ratio during this period (Muller et al. 2011; Hönekopp et al. 2007; Kowal et al. 2020; Richards et al. 2017). However, some studies reported the opposite pattern showing that 2D:4D ratio may change with age (Kyriakidis 2021).
Hand morphogenesis occurs between 6 and 14 weeks of gestation and it consists of three main stages: shape forming (from 6 to 10 weeks), the appearances of creases (from 10 to 13 weeks), and development of ridges (from weeks 13 onward). This process is regulated by many biochemical factors, such as protein Sonic hedgehog (Shh) and wingless-type mouse mammary tumor virus integration site family member 7a (Wnt-7a), which induces transcription of the factor LIM homeobox transcription factor that takes part in dorsalization of the limb bud (Lacroix et al. 1984).
Rodent studies investigating hand morphogenesis have shown that length of the fourth finger is determined by the balance of testosterone to estrogen during a small window in fetal development probable around 14th week of gestation. Androgen and estrogen receptor activity is greater in the development of digit 4 compared to digit 2. Sex steroids regulate a network of genes that are involved in chondrocyte proliferation which leads to the growth of digit 4 independently of sex. Higher levels of androgens simulate chondrocytes proliferation of the fourth finger and leads to an increased 2D:4D ratio and oppositely in the case of higher level of estrogen (Brown et al. 2002; Zheng and Cohn 2011). Beside the time of establishment of 2D:4D ratio, it is not clear whether 2D:4D proportion is changing from generation to generation.
The aim of this study was to evaluate the differences in digit ratio among Poles in three age groups: children, young adults and adults.
The data comes from three cohorts investigated in years 2015–2021 in Lodz (city in Central Poland, population 680,000). The information about individuals age was obtained via survey filled in by study participants or, if underaged, their parents.
Our cross-sectional study included 960 ethnically homogeneous healthy Caucasians (530 females and 430 males) aged between 6–79 years divided into three groups: children (6–13 years), young adults (18–29 years) and adults (30–79 years).
The youngest cohort was instigated in years 2015–2017 in randomly selected primary schools in Lodz and included 611 children (319 girls and 292 boys). All measurements were carried out by the employees of the Department of Anthropology of the University of Lodz.
Individuals in the 18–30 years age range were measured in Lodz between September 2020 and March 2021 by medical students (Medical University of Lodz) and by the employees of the Department of Anthropology of the University of Lodz. There were investigated 167 individuals (104 females and 63 males).
The oldest cohort, aged over 29 years, included 175 adults (101 females and 74 males) investigated between July 2020 and September 2020 in Lodz was carried out by the employees of the Department of Anthropology of the University of Lodz.
The direct measurements of the second and fourth finger were performed by qualified staff using a sliding calliper (Vernier calliper) with an accuracy of 0.001 m. Based on values of the fingers length, the 2D:4D index was calculated as a quotient of the length of the second digit and the fourth digit (mm).
The Consent of the Bioethics Committee at the Medical University of Lodz (RNN/374/19/KE and RNN/394/19/KE) and by the Ethical Commission at the University of Lodz (19/KBBN-UŁ/II/2016) were obtained. Written informed consent was obtained from all study participants or their parents in the case of children.
Due to lack of normal distribution of 2D:4D for right and left hand and age, the non-parametric tests were used.
The Kendall Tau correlation was applied to evaluate correlation between age and 2D:4D finger ratio for both hands.
Due to statistically significant differences between 2D:4D digit ratio for right and left hand (t=-2.106; p=0.035), further analyses were conducted separately for the right and left hand. However, among both sexes 2D:4D of left and right hand were positively correlated (females: r= 0.610; p<0.001; males: r=0.596; p<0.001).
The Mann Withney test was used to determine whether there were dimorphic differences in 2D:4D ratio of right and left hand with regards to age.
The Kruskal Wallis test (H) with Tukey’s post hoc tests were used to calculate differences between the three age groups.
The Cohen’s d values were calculated to calculate the effect size for each comparison.
All statistical analyses were performed using the Statistica ver. 13.0 software.
Although there were no significant age differences between males and females, there were dimorphic differences regarding 2D:4D (R) and 2D:4D (L) ratios. For example, females were characterized by higher 2D:4D (R) and 2D:4D (L) compared to males (Table 1).
| sex | variables | N | Mean | Median | Minimum | Maximum | Lower Quartile | Upper Quartile | Std.Dev. | Females vs Males | Coh-en’s d | |
| Z | p-value | |||||||||||
| Females | Age | 530 | 20.365 | 11.641 | 5.936 | 79.000 | 8.494 | 22.000 | 17.729 | 1.390 | 0.164 | 0.110 |
| Males | 430 | 18.479 | 11.374 | 5.919 | 79.000 | 8.404 | 21.000 | 16.584 | ||||
| Children F | Age | 319 | 9.334 | 9.012 | 5.936 | 13.303 | 7.726 | 11.036 | 1.941 | -1.278 | 0.201 | 0.102 |
| Children M | 292 | 9.534 | 9.163 | 5.919 | 13.148 | 7.866 | 11.442 | 1.994 | ||||
| Young adults F | Age | 104 | 21.356 | 21.000 | 19.000 | 28.000 | 20.000 | 22.000 | 1.751 | 0.469 | 0.639 | 0.074 |
| Young adults M | 63 | 21.508 | 21.000 | 19.000 | 29.000 | 20.000 | 23.000 | 2.341 | ||||
| Adults F | Age | 107 | 52.299 | 51.000 | 30.000 | 79.000 | 43.000 | 62.000 | 12.564 | 0.921 | 0.357 | 0.116 |
| Adults M | 75 | 50.760 | 47.00 | 30.000 | 79.000 | 40.000 | 64.000 | 13.946 | ||||
| Females | 2D:4D (R) | 530 | 0.986 | 0.986 | 0.881 | 1.095 | 0.966 | 1.000 | 0.033 | 4.985 | <0.001 | 0.333 |
| Males | 430 | 0.975 | 0.975 | 0.867 | 1.058 | 0.952 | 1.000 | 0.033 | ||||
| Females | 2D:4D (L) | 530 | 0.987 | 0.986 | 0.838 | 1.097 | 0.970 | 1.000 | 0.031 | 4.457 | <0.001 | 0.290 |
| Males | 430 | 0.978 | 0.978 | 0.883 | 1.092 | 0.961 | 1.000 | 0.031 | ||||
The 2D:4D digit ratios for the left hand were statistically significantly correlated with age both among females (Table 2) and males (Table 3). In the case of females, the 2D:4D digit ratios for the right hand were also statistically significantly correlated with age (Table 2). Correlation between digit ratio and age within each of the age groups was non-significant (Table 2 and 3).
| Groups | N | 2D:4D R & age | 2D:4D L & age | ||||
| Kendall Tau (τ) | Z | p | Kendall Tau (τ) | Z | p | ||
| Females (total) | 530 | 0.082 | 2.834 | 0.005 | 0.137 | 4.716 | <0.001 |
| children | 319 | 0.032 | 0.857 | 0.392 | 0.066 | 1.757 | 0.079 |
| young adults | 104 | 0.048 | 0.720 | 0.472 | 0.050 | 0.735 | 0.451 |
| adults | 107 | 0.027 | 0.413 | 0.679 | 0.049 | 0.748 | 0.454 |
| Groups | N | 2D:4D R & age | 2D:4D L & age | ||||
| Kendall Tau (τ) | Z | p | Kendall Tau (τ) | Z | p | ||
| Males (total) | 430 | 0.039 | 1.221 | 0.222 | 0.083 | 2.579 | 0.010 |
| children | 292 | 0.027 | 0.710 | 0.480 | 0.043 | 1.092 | 0.275 |
| young adults | 63 | -0.028 | -0.329 | 0.742 | -0.095 | -1.103 | 0.270 |
| adults | 75 | -0.145 | -1.842 | 0.065 | 0.022 | 0.282 | 0.778 |
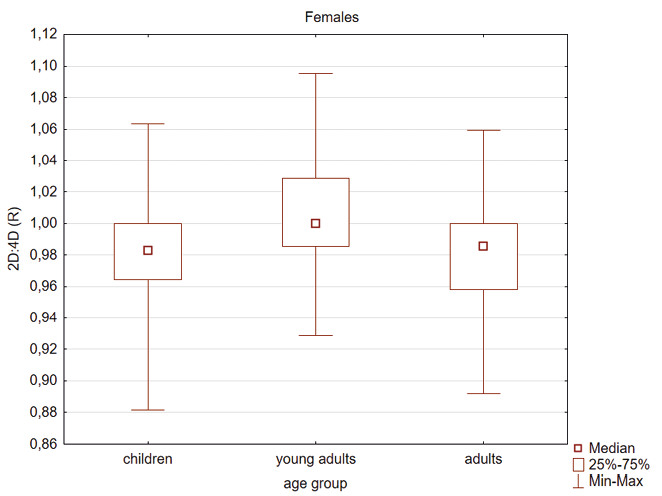
Kruskal Wallis test showed that young adult females had higher 2D:4D (R) compared to children and adults (Table 4, Figure 1). In the male group the 2D:4D (R) was higher in young adults than in children (Table 4, Figure 2). In the case of 2D:4D (L) there were the following differences according to age groups: female children had lower digit ratio (L) than young adults and adults (Table 4, Figure 3) and young adults males had higher digit ratio (L) compared to children (Table 4, Figure 4).
| Age group | Variables | N | Mean | Median | Minimum | Maximum | Lower Quartile | Upper Quartile | Std.Dev. | St. Error | H | p |
| Females | ||||||||||||
| children | 2D:4D (R) | 319 | 0.982 | 0.983 | 0.881 | 1.063 | 0.964 | 1.000 | 0.030 | 0.002 | 36.383 | <0.0011 |
| young adults | 104 | 1.004 | 1.000 | 0.929 | 1.095 | 0.985 | 1.028 | 0.033 | 0.003 | |||
| adults | 107 | 0.982 | 0.986 | 0.892 | 1.060 | 0.958 | 1.000 | 0.035 | 0.003 | |||
| Males | ||||||||||||
| children | 2D:4D (R) | 292 | 0.973 | 0.976 | 0.867 | 1.058 | 0.950 | 1.000 | 0.033 | 0.002 | 6.053 | 0.0492 |
| young adults | 63 | 0.986 | 0.986 | 0.922 | 1.054 | 0.961 | 1.013 | 0.032 | 0.004 | |||
| adults | 75 | 0.974 | 0.974 | 0.893 | 1.042 | 0.949 | 1.000 | 0.035 | 0.004 | |||
| Females | ||||||||||||
| children | 2D:4D (L) | 319 | 0.981 | 0.983 | 0.838 | 1.058 | 0.967 | 1.000 | 0.029 | 0.002 | 21.338 | p<0.0013 |
| young adults | 104 | 0.998 | 1.000 | 0.920 | 1.097 | 0.972 | 1.016 | 0.033 | 0.003 | |||
| adults | 107 | 0.994 | 0.986 | 0.910 | 1.057 | 0.971 | 1.015 | 0.033 | 0.003 | |||
| Males | ||||||||||||
| children | 2D:4D (L) | 292 | 0.975 | 0.977 | 0.883 | 1.053 | 0.956 | 1.000 | 0.030 | 0.002 | 9.097 | 0.0114 |
| young adults | 63 | 0.989 | 0.986 | 0.938 | 1.056 | 0.971 | 1.014 | 0.029 | 0.004 | |||
| adults | 75 | 0.982 | 0.977 | 0.902 | 1.092 | 0.960 | 1.000 | 0.034 | 0.004 | |||
Statistically significant post hoc tests: | 3 2D:4D (L) Females |

Fig. 1. Differentiation of 2D: 4D finger ratio of the right hand between the three age groups. Statistically significant effects: young adults and children (p<0.001) and young adults and adults (p<0.001)
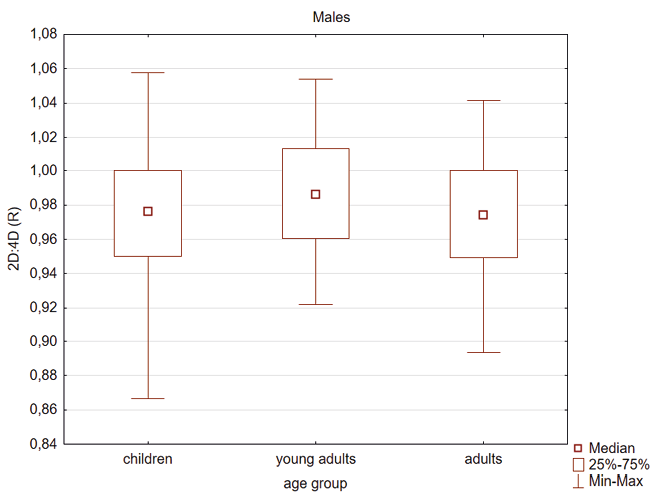
Fig. 2. Differentiation of 2D: 4D finger ratio of the left hand between the three age groups among males. Statistically significant effects: young adults and children (p=0.046)
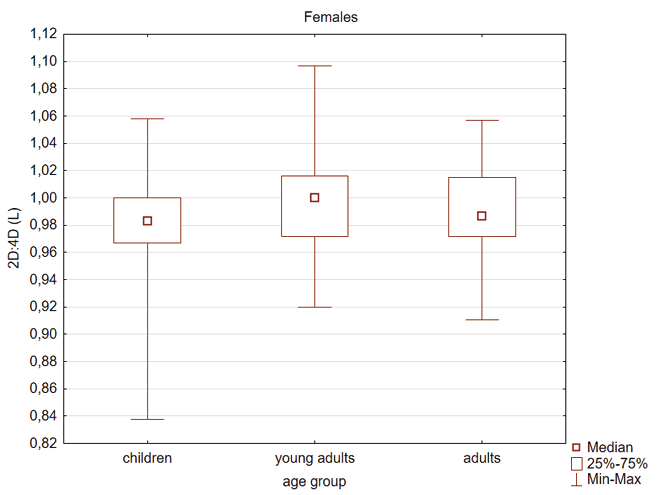
Fig. 3. Differentiation of 2D: 4D finger ratio of the left hand between the three age groups among females. Statistically significant effects: adults and children (p=0.005) and young adults and children (p<0.001)

Fig. 4. Differentiation of 2D: 4D finger ratio of the left hand between the three age groups among males. Statistically significant effects: young adults and children (p=0.011)
Although the relationship between the 2D:4D digit ratio and biology, behaviour and health has been widely investigated (Pruszkowska-Przybylska et al. 2008; Pruszkowska-Przybylska et al. 2021; Kasielska-Trojan et al. 2020; Sitek et al. 2018; Kobus et al. 2021) the association between 2D:4D and age is poorly understood mainly because studies have reported inconsistent results.
Regardless of the ethnic group, some changes in the 2D:4D digit ratio may occur during the prenatal period, the first two years after birth, and in later life (Butovskaya et al. 2021; Knickmeyer et al. 2011). The results of our study show that there are generation differences in the 2D:4D digit ratio for both hands between children and young adults. Older individuals seem to have higher digit ratios independently of sex possibly because there is an estrogen impact on digit development observed during late adolescence. In addition, Kyriakidis et al. (2021) reported that Greeks aged 38–63 years old had significantly higher 2D:4D ratio compared to a group aged <38 and ≥64 years old. In contrary, a study conducted by Kobus et al. (2021) showed no statistically significant correlation between 2D:4D observed across generations in a group of Poles aged 18–76 years. Similarly, Manning and Fink (2018) reported insignificant influence of age and no interaction effect of age and sex on 2D:4D in children or adults. The relationship between 2D:4D digit ratio and age has been also investigated in several longitudinal studies suggesting that 2D:4D digit ratio is not unstable in children and adolescents (Trivers et al. 2020; McIntyre et al. 2006; Trivers et al. 2006; Králík et al. 2017; Körner et al. 2020; Guo et al. 2021). One study by Richards et al. (2017) showed that 2D:4D among adult males was negatively correlated with age. The results of this study also show a negative, although non-significant, trend among adult males regarding the association of 2D:4D (right hand) with age. It is possible that chondrocytes are regulated during adulthood by testosterone, which, compared to children, is at a higher and more stable level in adults.
The time of the hand morphogenesis corresponds to the prenatal sex hormone exposure although there is no evidence that proportion of second and fourth finger remains equal during the entire ontogenesis. Moreover, longitudinal cohort study by ethnicity is needed to determine possible fluctuating changes of 2D:4D ratio.
We underline that confirmation of the correlation between 2D:4D digit ratio and age does not exclude the 2D:4D value as a determinant of the influence of sex hormones in prenatal development. However, each analysis should always be started by checking the relationship of the finger index in a given studied group with age and, if there is such a relationship, control for the influence of age on the value of this indicator while examining other effects.
Developing research in this area is necessary to create universal correction tool for each investigated group for 2D:4D research.
The limitation of the study might be not equal number of individuals from each stage of the ontogenesis.
Due to cross sectional nature of the study the effect that was observed could be a secular effect within age sections of a population.
Another limitation of the study might be a wide range of age that could also have biological implication. For instance, the pattern of relationship with age might have higher order differences between younger and older age groups. Thus, longitudinal studies that include the same individuals are needed to provide information to supplement our findings.
There might be a possible association between digit ratio and age, and direction of the correlation might be related to the phase of the ontogenesis. The results of our study suggest that controlling the influence of age on the 2D:4D digit ratio value while examining other effects is important.
Conflict of interest
The Authors declare no conflict of interests.
Authors’ contributions
P.P.-P. designed the study, collected the material, analyzed the data, and prepared the draft and final version of the manuscript. M.K. collected the material and participated in preparing the draft of the manuscript. E.Ż. participated in preparing the manuscript and provided critical comments on the manuscript. I.R. and M.P. collected the material. W.K. participating in the study designing A.S. designed the study, collected the material, participated in preparing the manuscript and provided critical comments on the manuscript.
Brown WM, Finn CJ, Breedlove SM. 2022. Sexual dimorphism in digit-length ratios of laboratory mice. Anat Rec 267:231–234. https://doi.org/10.1002/ar.10108
Butovskaya M, Burkova V, Apalkova Y. et al. 2021. Sex, population origin, age and average digit length as predictors of digit ratio in three large world populations. Sci Rep 11:8157. https://doi.org/10.1038/s41598-021-87394-6
de Sanctis V, Soliman AT, Elsedfy H, Soliman N, Elalaily R, Di Maio S. Is the Second to Fourth Digit Ratio (2D:4D) a Biomarker of Sex-Steroids Activity? 2017. Pediatr Endocrinol Rev 14(4):378–386. https://doi.org/10.17458/per.vol14.2017.SSE.SexSteroids
Guo J, Wu C, Zhang J, Li W, Lv S, Lu D, Qi X, Feng C, Liang W, Chang X, Zhang Y, Xu H, Cao Y, Wang G, Zhou Z. 2020. Prenatal exposure to multiple phenolic compounds, fetal reproductive hormones, and the second to fourth digit ratio of children aged 10 years in a prospective birth cohort. Chemosphere 263:127877. https://doi.org/10.1016/j.chemosphere.2020.127877
Hönekopp J, Bartholdt L, Beier L, Liebert A. 2007. Second to fourth digit length ratio (2D:4D) and adult sex hormone levels: new data and a meta-analytic review. Psychoneuroendocrinology 32:313–321 https://doi.org/10.1016/j.psyneuen.2007.01.007
Kasielska-Trojan A, Manning JT, Antczak A, Dutkowska A, Kuczyński W, Sitek A, Antoszewski B. 2020. Digit ratio (2D: 4D) in women and men with lung cancer. Scientific Reports 10(1):1–8. https://doi.org/10.1038/s41598-020-68239-0
Knickmeyer RC, Woolson S, Hamer RM, Konneker T, Gilmore JH. 2011. 2D:4D ratios in the first 2 years of life: Stability and relation to testosterone exposure and sensitivity. Horm Behav 60(3):256–263. https://doi.org/10.1016/j.yhbeh.2011.05.009
Kobus M, Sitek A, Rosset I, Pruszkowska-Przybylska P, Żądzińska E. 2021. Association of prenatal sex steroid exposure estimated by the digit ratio (2D:4D) with birth weight, BMI and muscle strength in 6-to 13-year-old Polish children. Plos One 16(10):e0258179. https://doi.org/10.1371/journal.pone.0258179
Kobus M, Sitek A, Antoszewski B, Rożniecki J, Pełka J, Żądzińska E. 2021. Prenatal oestrogen-testosterone balance as a risk factor of migraine in adults. J Headache Pain 22:119. https://doi.org/10.1186/s10194-021-01326-3
Kowal M, Sorokowski P, Żelaźniewicz A, et al. 2020. No relationship between the digit ratios (2D:4D) and salivary testosterone change: Study on men under an acute exercise. Sci Rep 10:10068. https://doi.org/10.1038/s41598-020-66915-9
Körner LM, Schaper ML, Pause BM, Heil M. 2020. Parent-Reports of Sex-Typed Play Preference in Preschool Children: Relationships to 2D:4D Digit Ratio and Older Siblings’ Sex. Arch Sex Behav 49(7):2715–2724. https://doi.org/10.1007/s10508-020-01662-6
Králík M, Ingrová P, Kozieł S, Hupková A, Klíma O. 2017. Overall trends vs. individual trajectories in the second-to-fourth digit (2D:4D) and metacarpal (2M:4M) ratios during puberty andadolescence. Am J Phys Anthropol 162(4):641–656. https://doi.org/10.1002/ajpa.23153
Kyriakidis I. 2021. Data regarding 2D:4D and other digit ratios in Greek population. Data in brief 34:106724. https://doi.org/10.1016/j.dib.2021.106724
Lacroix B, Wolff-Quenot MJ, Haffen K. 1984. Early human hand morphology: an estimation of fetal age. Early Hum Dev 9(2):127–36. https://doi.org/10.1016/0378-3782(84)90093-8
Lutchmaya S, Baron-Cohen S, Raggatt P, Knickmeyer R, Manning JT. 2004. 2nd to 4th digit ratios, fetal testosterone and estradiol. Early Hum Dev 77(1–2):23–28. https://doi.org/10.1016/j.earlhumdev.2003.12.002
Manning J, Kilduff L, Cook C, Crewther B, Fink B. 2014a. Digit ratio (2D:4D): a biomarker for prenatal sex steroids and adult sex steroids in challenge situations. Front Endocrinol 5:9. https://doi.org/10.3389/fendo.2014.00009
Manning JT, Fink B. 2018. Sexual dimorphism in the ontogeny of second (2D) and fourth (4D) digit lengths, and digit ratio (2D:4D). Am J Hum Biol 30(4):e23138. https://doi.org/10.1002/ajhb.23138
McIntyre MH, Cohn BA, Ellison PT. 2006. Sex dimorphism in digital formulae of children. Am J Phys Anthropol 129(1):143–50. https://doi.org/10.1002/ajpa.20240
Muller DC, Giles GG, Bassett J. et al. 2011. Second to fourth digit ratio (2D:4D) and concentrations of circulating sex hormones in adulthood. Reprod Biol Endocrinol 9:57. https://doi.org/10.1186/1477-7827-9-57
Pruszkowska-Przybylska P, Sitek A, Rosset I, Sobalska-Kwapis M, Słomka M, Strapagiel D, Żądzińska E. 2018. Association of the 2D: 4D digit ratio with body composition among the Polish children aged 6–13 years. Early Hum Dev 124:26–32. https://doi.org/10.1016/j.earlhumdev.2018.08.001
Pruszkowska-Przybylska P, Sitek A, Rosset I, Sobalska-Kwapis M, Słomka M, Strapagiel D, Żądzińska E, Morling N. 2021. Associations between second to fourth digit ratio, cortisol, vitamin D, and body composition among Polish children. Scientific Reports 11(1):1–9. https://doi.org/10.1038/s41598-021-86521-7
Richards G, Bellin W, Davies W. 2017. Familial digit ratio (2D:4D) associations in a general population sample from Wales. Early Hum Dev 112:14–19. https://doi.org/10.1016/j.earlhumdev.2017.06.006
Sammer DM, Chung KC. 2009. Congenital hand differences: embryology and classification. Hand Clin 25(2):151–6. https://doi.org/10.1016/j.hcl.2009.02.002
Sitek A, Kozieł S, Kasielska-Trojan A, Antoszewski B. 2018. Do skin and hair pigmentation in prepubertal and early pubertal stages correlate with 2D:4D?. Am J Hum Biol 30(6):e12631. https://doi.org/10.1002/ajhb.23183
Trivers R, Manning J, Jacobson A. 2006. A longitudinal study of digit ratio (2D:4D) and other finger ratios in Jamaican children. Horm Behav 49(2):150–6. https://doi.org/10.1016/j.yhbeh.2005.05.023
Trivers R, Jacobson A, Manning JT. 2020. Radiographic digit ratios (2D:4D) of Afro-Caribbean children: Comparisons with published data from white children. Early Hum Dev 146:105072. https://doi.org/10.1016/j.earlhumdev.2020.105072
Zheng Z, Cohn MJ. 2011. Developmental basis of sexually dimorphic digit ratios. Proc Natl Acad Sci 108(39):16289–16294. https://doi.org/10.1073/pnas.1108312108

